RES | PM | 01.08.2023
For the production of polyethylene, which is found for example in film products, ethylene is needed as a monomer. The production of ethylene today is mainly based on fossil raw materials. Steamcracking is used to convert the fossil raw materials into short-chain molecules molecules such as ethylene or propylene. In this process, as well as the end-of-life use of the final product, generates significant CO2 emissions. However, there are new, sustainable process routes for the production of ethylene, which can be extended by a further promising route through the work at the Professorship of Regenerative Energy Systems.
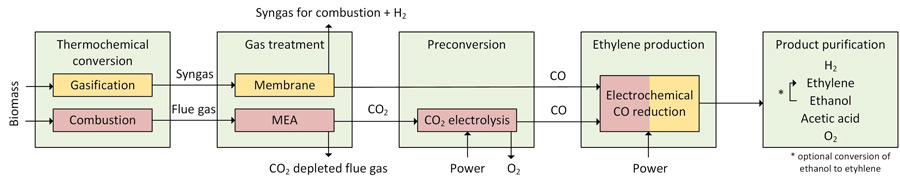
Daniel Klüh, together with a team of Finnish scientists at Lappeenranta University of Technology (Harri Nieminen, Kristian Melin, Arto Laari and Tuomas Koiranen) developed and assessed a new production route. As shown in the figure (yellow boxes), carbon monoxide is obtained from the gasification of biomass residues and is converted electrochemically. In addition to ethylene, other valuable products such as oxygen, hydrogen, ethanol and acetic and acetic acid. This new route contrasts with a route that has often been described in science with CO2 as the starting material (red boxes, hereafter referred to as the comparative process).
Based on a techno-economic evaluation, it was shown that the production costs for ethylene via the electrochemical reduction of carbon monoxide are approximately three times higher than the usual market prices. However, the cost price of the comparative process is 4 to 6 times higher. The energy efficiency of the process could be improved to 42-43% compared to 28% for the comparative process. In addition, CO2 emissions can be reduced more significantly compared to the comparative process. Thus, when green electricity is used to operate the plant, a wide range of low-emission chemical feedstocks can be produced. Furthermore, it was shown that further optimization of selectivity to ethylene does not achieve significantly lower costs or higher efficiencies. Using an established process to convert ethanol to ethylene, a higher ethylene yield can still be achieved with a less selective process.
Further information can be found in the following publication (open access): https://doi.org/10.3389/fenrg.2023.1129076

